|
How many of the following molecules are polar? BrCl3 CS2 SiF4 SO3. Eg=trigonal planar, mg=trigonal planar, nonpolar. Determine the electron geometry (eg), molecular geometry (mg), and polarity of SO3. Picasa Mac Download Free Adobe Cs2 Download Full Version Mac Os X Flash Player 10.1 Mac Download Voice Recorder Software Free Download For Mac Adobe Illustrator Mac Free Download Adobe Download For Mac No Cd Adobe Photoshop For Mac Free Download Full Version Keygen Adobe Photoshop Free Trusted Download For Mac The first time, we are providing software for engineering technology.Adobe illustrator cc 2018 download full version for.Adobe Photoshop CS2 free download with serial number. Soft Gudam is a largest storage of pc software and games for free download. Write out each of their lewis. Log in to reply to the answers Post halee8202.
Adobe Illustrator Cs2 For Mac Adobe Illustrator(A) C7H16, C5H12 (B) CCl4, CBr4 (C) H2O, H2S (D) CH3CH2OH, CH3—O—CH3 Select the pair of compounds in which the substance with the higher vapor pressure at a given temperature is listed first. It can be downloaded for free without any trouble in j That’s all that you need to You can download Adobe Illustrator CS2 latest version free complete standalone offline file for free. Step 5: Copy and paste the serial number that is beside the download to save it and then use it for activating the CS2 version of Photoshop. 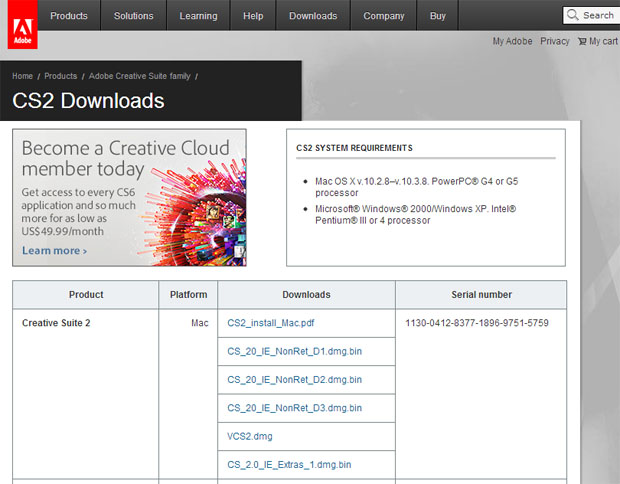
Non-Polar Molecule Definition: A non-polar molecule is one that the electrons are distributed more symmetrically and thus does not have an abundance of charges at the opposite sides. Which of the isomers of C 6 H 3 F 3 has the highest boiling point? 10.g 10.h Non-polar Refer to question 10 above. Chemistry “A sample of a nonpolar molecules capable of only dispersion forces can have a higher boiling point the a sample of a polar molecules capable of hydrogen bonding. Asked by Anonymous on April 5, 2016. Which of the following molecules is/are nonpolar? H2S O3 COS C2H2 SO2 SiF4 PF3 HCN NH3 CS2 HN3 SO3 CF4 BF3 H2O N2O AsH3. Is CS2 polar or nonpolar? Join Yahoo Answers and get 100 points today. Solution Update 2: thanks alot. The chemical composition of this substance is such that it consists of 1 carbon and 2 sulfur atoms. Ring 1 Instead of having polar bonds, the molecule is nonpolar. This makes a total of 16 valence electrons of the CS2 molecule. When activated you will switch on Positive and. This event needs to be activated from the Season 1 > Polarity Switch page. The Polarity Switch event is a currently running event in The Division 2. The arrangement or geometry of the atoms in some molecules is such that one end of the molecule has a positive electrical charge and the other side has a negative charge. By Ron Kurtus (revised 19 September 2016) Molecules can be grouped as polar or non-polar molecules.Some molecules are in between the two. If the electrons are shared equally between the atoms then its a non-polar covalent bond. This is determined with the concept of electro-negativity. Covalent bond between the elements can be either polar or non-polar. Chemical bond polarity is the concept that explains the property of sharing an electron between two elements. What about sulfur dioxide, is it polar or nonpolar? Sulfur dioxide is considered a polar molecule. For instance, water is a polar molecule while carbon dioxide is a nonpolar molecule. Carbon dioxide does not form a non-polar molecular since its geometry is linear. An example of this scenario is carbon dioxide (CO 2). An important fact that one needs to take note of is that not every molecule with polar bonds is a polar molecule. Out of these four compounds, CS2 is the only one that does not have polarity differences causing deviations from ideal bond angles. CS 2 - Carbon Disulfide: First draw the Lewis dot structure: Electron geometry: linear. Zsnes emulator for macCarbon tetrachloride (CCl4) is also nonpolar but is made of larger molecules that are more strongly affected by London dispersion forces. The molecule carbon disulfide (CS2) is nonpolar and has only London dispersion forces between the molecules. De- tection of the latter through the degree of polar- ization of the fluorescence, first observed by PHOTOSELECTION OF CS2 VAPOR Photoselection by excitation with a beam of polarized light is well-known for polyz+_tomic molecules in condensed media , and for dia- tomic molecules in the gas phase [ll, 131. Sempre que quiser saber se uma molécula é polar ou apolar lembre-se da pilha e analise a fórmula estrural da molécula (não esquecer de também analisar a polaridade das ligações). CO 2 is nonpolar because the two polar bonds are equal and opposite so cancel out H 2O is polar because the bonds are not opposite and don’t. The C–C bonds in C 3 H 8 _nonpolar covalent_ ii. Polar bond occurs between a more electronegative atom and a electropostive atom.molecules with such polar bonds are called as polar molecules.ex-H2O,NaCl,MgO nonpolar bond occurs between to like atoms or between any other atoms.both the atoms should have the same electronegativity value.ex-O2,H2,F2Polar molecules happen when there is an electronegativity difference among the bonded atoms. For the best answers, search on this site. HCN Im stuck between C and D C is a tetrahedral which has no net dipole so is non polar, and D is linear which Choose the compound below that contains at least one polar covalent bond, but is nonpolar a. O carbono é de família IVA, necessitando de 4 ligações. Example, Carbon-Hydrogen or Hydrogen-Hydrogen. If it is less than 0.5, then it is a non-polar covalent bond. Polar and Nonpolar Covalent Bonds Polar and Nonpolar Molecules Polar MoleculesElectronegativity difference is a great tool to predict the nature of bond whether it is polar, non-polar or ionic. Note the number of electron regions around the central atom, and of these which are bonding or lone pairs (non-bonding pairs) Step 2: Use this info to determine the 3D. Step 1: Draw the Lewis structure. (d) predict the molecular shapes for CS32− and CS2 and explain how you arrived at your.These are problems using 3D molecules run in the application Jmol to help you visualize the molecule to determine if it is polar or non-polar. Polar Pattern.4 years ago. In the C-H bond, carbon pulls the negative charge to its side and in C-Cl bonds, chlorine pulls the negative charge (electrons) to its side as chlorine is more electronegative than carbon.CS-2. Therefore C-H and C-Cl bonds are polar. Poole, in The Essence of Chromatography, 2003. CS2 By signing up, you'll get thousands of step-by-step solutions. Answer to: Of the molecules below, only is polar. Note the number of electron regions around the central atom, and of these which are bonding or lone pairs (non-bonding pairs) Step 2: Use this info to determine the 3D. Step 1: Draw the Lewis structure.
0 Comments
Leave a Reply. |
AuthorPeter ArchivesCategories |
 RSS Feed
RSS Feed
